POSTS
31/01/2023
Regolamento (EU) n. 536/2014 sulle sperimentazioni cliniche
22/11/2022 Market Surveillance of Medical Devices
01/09/2022 LILLA LINE INTERNATIONAL LICENSING & ACQUISITIONS
14/01/2022 RE-START 2022
03/06/2021 COVID AS A SAE IN CLINICAL INVESTIGATIONS
09/02/2021 Clinical Evidence Management
30/11/2020 SELF WELLBEING | body | mind | work | world
26/10/2020 QUALITY STANDARDIZATION | SERVICE SUSTAINABILITY | PEOPLE WELLBEING
06/07/2020 #SDG 2 Treatment of Anaemia | Food Supplement RUBENFLUX®
08/06/2020 REGULATORY COMPLIANCE SERVICES | Smart Wellbeing Modulations
Regolamento (EU) n. 536/2014 sulle sperimentazioni cliniche
31/01/2023

Dopo il periodo di transizione, dalla data odierna 31 gennaio 2023 tutte le procedure di autorizzazione della sperimentazione clinica devono obbligatoriamente essere richieste seguendo quanto contenuto nel nuovo Regolamento (EU) n. 536/2014 sulle sperimentazioni cliniche di medicinali per uso umano.
L’obiettivo del Regolamento è l’armonizzazione della disciplina europea in materia di sperimentazioni cliniche, per garantire standard di sicurezza elevati per i pazienti/soggetti e per rafforzare la collaborazione degli Stati Membri. Il Regolamento introduce una procedura armonizzata per la valutazione delle domande di autorizzazione, articolata in tre fasi: convalida, valutazione; e decisione.
Per ottenere l’autorizzazione, il promotore della sperimentazione deve presentare mediante il portale CTIS (Clinical Trials Information System) di EMA, un fascicolo di domanda agli Stati membri interessati in cui intende condurre la sperimentazione stessa, proponendo uno Stato membro relatore (Reporting Member State).
Restiamo a disposizione per fornire ulteriori informazioni del caso e per supportare i Clienti in tutti gli step necessari per ottenere riscontro positivo da parte di EMA.
L’obiettivo del Regolamento è l’armonizzazione della disciplina europea in materia di sperimentazioni cliniche, per garantire standard di sicurezza elevati per i pazienti/soggetti e per rafforzare la collaborazione degli Stati Membri. Il Regolamento introduce una procedura armonizzata per la valutazione delle domande di autorizzazione, articolata in tre fasi: convalida, valutazione; e decisione.
Per ottenere l’autorizzazione, il promotore della sperimentazione deve presentare mediante il portale CTIS (Clinical Trials Information System) di EMA, un fascicolo di domanda agli Stati membri interessati in cui intende condurre la sperimentazione stessa, proponendo uno Stato membro relatore (Reporting Member State).
Restiamo a disposizione per fornire ulteriori informazioni del caso e per supportare i Clienti in tutti gli step necessari per ottenere riscontro positivo da parte di EMA.
Image by Gerd Altmann from Pixabay
#blymum #clinicaltrials #regulatoryaffairs
#blymum #clinicaltrials #regulatoryaffairs
Market Surveillance of Medical Devices
22/11/2022

All manufacturers of medical devices (MDs) must minimize the risk posed by their MDs and ensure patient safety. Post-market surveillance is thus mandatory. According to the new MDR (EU 2017/745), systematic monitoring is needed following a precise frequency.
For Class I, Is, Im, and Ir MDs, every 4 years at a minimum (the submission to notified bodies is not required). For Class II MDs a PSUR (Periodic Safety Update report) is required every two years at a minimum (upon request the report must be available to the relevant notified body and competent health authority).
For Class IIb MDs a PSUR is required annually (upon request the report must be available to the relevant notified body and competent health authority).
For Class III MDs and implants a PSUR is required to be submitted annually using the EUDAMED system.
We can for sure assist all manufacturers in such activities; do not hesitate to contact us at info@blymum.com
Image: Oleg Gamulinskiy from Pixabay
For Class I, Is, Im, and Ir MDs, every 4 years at a minimum (the submission to notified bodies is not required). For Class II MDs a PSUR (Periodic Safety Update report) is required every two years at a minimum (upon request the report must be available to the relevant notified body and competent health authority).
For Class IIb MDs a PSUR is required annually (upon request the report must be available to the relevant notified body and competent health authority).
For Class III MDs and implants a PSUR is required to be submitted annually using the EUDAMED system.
We can for sure assist all manufacturers in such activities; do not hesitate to contact us at info@blymum.com
Image: Oleg Gamulinskiy from Pixabay
LILLA LINE INTERNATIONAL LICENSING & ACQUISITIONS
01/09/2022
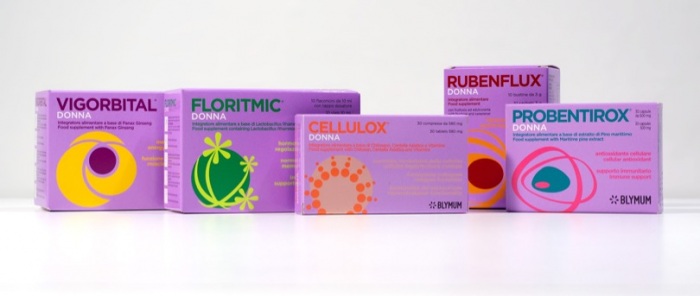
BLYMUM is proposing the LILLA LINE whole portfolio or eventually single products for international licensing and/or for definitive purchase. In case of products/portfolio acquisition, the product’s formula dossier, product samples, literature dossier, IP transfer (if available), testing dossier (if available), and supporting marketing and training contents are delivered.
For more information please contact info@blymum.com
RE-START 2022
14/01/2022

We can help You with innovation in 2022!
WORKPLACE WELLBEING
First of all, with the LIVELLA® Self Wellbeing Management project, dedicated to improving individual prosperity and comfort in the four spheres Body, Mind, Work and World, with tangible benefits both for Your people and for Your Company.
COLLABORATIVE INNOVATION
We support You within Collaborative Innovation processes, following ISO 56002 and 56003, through smart working on visual collaboration platforms (i.e., Mural, Miro), and personalized mentoring aimed at enhancing individual and group skills of Your human resources.
VALUE PROPOSITION MODELING
If you have to manage structural interventions of your business system, we also point out the BUSINESS MODEL based INNOVATION method (CINNAM BMI-ALDEHYDE MBS®) that greatly facilitates the management of changes at all levels, from strategic development of the company/brand to the formulation of frontline value propositions of commercial and technical offers for the market.
For all forms of collaboration, it is possible to interact in presence and/or remotely, always with a mindset of sustainability, wellbeing, smart working and creativity.
For more information and references contact: merlinova@blymum.com
Image courtesy of Gerd Altmann from Pixabay
WORKPLACE WELLBEING
First of all, with the LIVELLA® Self Wellbeing Management project, dedicated to improving individual prosperity and comfort in the four spheres Body, Mind, Work and World, with tangible benefits both for Your people and for Your Company.
COLLABORATIVE INNOVATION
We support You within Collaborative Innovation processes, following ISO 56002 and 56003, through smart working on visual collaboration platforms (i.e., Mural, Miro), and personalized mentoring aimed at enhancing individual and group skills of Your human resources.
VALUE PROPOSITION MODELING
If you have to manage structural interventions of your business system, we also point out the BUSINESS MODEL based INNOVATION method (CINNAM BMI-ALDEHYDE MBS®) that greatly facilitates the management of changes at all levels, from strategic development of the company/brand to the formulation of frontline value propositions of commercial and technical offers for the market.
For all forms of collaboration, it is possible to interact in presence and/or remotely, always with a mindset of sustainability, wellbeing, smart working and creativity.
For more information and references contact: merlinova@blymum.com
Image courtesy of Gerd Altmann from Pixabay
COVID AS A SAE IN CLINICAL INVESTIGATIONS
03/06/2021

SARS-CoV-2 aka Covid-19 is an untoward medical occurrence, unintended disease or injury, or untoward clinical sign (including abnormal laboratory findings) which is currently affecting some of the subjects enrolled in clinical studies. It is a Serious Adverse Event (SAE) that can lead to death or serious deterioration in the health of subjects, their families, or other persons.
We are currently monitoring a Randomized Double Blind Placebo Controlled Clinical Study to evaluate the efficacy of a specific Medical Device. Because of Covid-19 we have registered a statistically significant increase of drop-outs, dangerously approaching the threshold of subjects that we have set as the drop-out limit for the termination of the clinical investigation on statistical grounds.
The teaching is clear: during SARS-CoV-2 Pandemic, while defining the statistical design, the methods and the analytical procedures of a new clinical investigation, pay higher attention to the sample size, since it can be conditioned by further unpredictable occurrences. You will avoid additional enrollments, extra amendments to the CIP and preserve the robustness of the statistical results within previewed deadlines.
Image by Gerd Altmann from Pixabay
We are currently monitoring a Randomized Double Blind Placebo Controlled Clinical Study to evaluate the efficacy of a specific Medical Device. Because of Covid-19 we have registered a statistically significant increase of drop-outs, dangerously approaching the threshold of subjects that we have set as the drop-out limit for the termination of the clinical investigation on statistical grounds.
The teaching is clear: during SARS-CoV-2 Pandemic, while defining the statistical design, the methods and the analytical procedures of a new clinical investigation, pay higher attention to the sample size, since it can be conditioned by further unpredictable occurrences. You will avoid additional enrollments, extra amendments to the CIP and preserve the robustness of the statistical results within previewed deadlines.
Image by Gerd Altmann from Pixabay
Clinical Evidence Management
09/02/2021

Starting from May 2021, a significant change in legislation on medical devices (Regulation 2017/745) will reinforce the requirements for clinical evaluation and place more emphasis on a life-cycle approach to safety backed up by clinical data. Act now to be ready on time!
Our services pertaining to the MDR are proposed as BLYMUM Clinical Evidence Management (B-CEM). They are aimed at :
For more information:
Clinical Evidence Management
Image background: Gerd Altmann
Our services pertaining to the MDR are proposed as BLYMUM Clinical Evidence Management (B-CEM). They are aimed at :
- casting partners for clinical investigations, in order to ensure the collaboration with the most appropriate and internationally recognised scientists and experts from primary academic and research Institutions,
- ground-working, supporting You throughout the scientific documentation assessment, regulatory compliance, investigational site assessment, and risk management (ISO 14971),
- planning clinical investigations, monitoring activities, and post market clinical follow-up; composing, according to the MDR, instructions, the clinical investigation plan (CIP) monitoring plan (MP) and clinical follow-up plan CFP), and
- monitoring and reporting data according to the monitoring plan.
For more information:
Clinical Evidence Management
Image background: Gerd Altmann
SELF WELLBEING | body | mind | work | world
30/11/2020

Several researches, such as the one from Euromonitor International (https://go.euromonitor.com/white-paper-ch-2020-health-by-generation.html ) confirm the continuous and global increase in the fragility and discomfort of people. Being aware of the several individual difficulties and their impact on personal well-being, we decided to rethink the settings of our Livella® program dedicated to the management of Self-Wellbeing.
The Livella® philosophy and methodology, inspired by the physiological modulation theory (https://www.blymum.com/contents/files/PAPER-PH.MODULATORS-CORNELLI.pdf ), is currently focused on improving physical (BODY) and mental (MIND) health. However, work and social relationships, in difficult contexts as it is today for many, become truly crucial for existence and personal comfort and prosperity.
Consequently, from now on, Livella® Self-Wellbeing professional consulting and mentoring integrates also Self-Management, Self-Marketing and Self-Innovation tracks for the management of the working sphere and interpersonal relationships. Livella®, our approach to the balance management of the human being, will be defined by the proportions among BODY, MIND, WORK and WORLD dimensions, with tangible benefits for both people, companies and the market.
The Livella® philosophy and methodology, inspired by the physiological modulation theory (https://www.blymum.com/contents/files/PAPER-PH.MODULATORS-CORNELLI.pdf ), is currently focused on improving physical (BODY) and mental (MIND) health. However, work and social relationships, in difficult contexts as it is today for many, become truly crucial for existence and personal comfort and prosperity.
Consequently, from now on, Livella® Self-Wellbeing professional consulting and mentoring integrates also Self-Management, Self-Marketing and Self-Innovation tracks for the management of the working sphere and interpersonal relationships. Livella®, our approach to the balance management of the human being, will be defined by the proportions among BODY, MIND, WORK and WORLD dimensions, with tangible benefits for both people, companies and the market.
Image courtesy of Gerd Altmann from Pixabay
QUALITY STANDARDIZATION | SERVICE SUSTAINABILITY | PEOPLE WELLBEING
26/10/2020

Blymum supports explicitly the Sustainable Development Goals of the United Nations and we fully adhere to the 3rd SDG: Good Health and Wellness for People. We provide services to pharmaceutical, food, healthcare and other clients and, through our professional activities, we concretely contribute to people’s health, physical and psychological wellbeing, longevity and beauty.
All customers and consumers today ask for greater safety and more guarantees. For the sake of satisfying these specific requests and ensuring the correctness, sustainability and efficiency of our working processes, we observe in particular the following ISO Standards: ISO 14155 - Clinical Investigation of Medical Devices for Human Subjects; ISO 22000 - Food Safety Management; ISO 45001 - Occupational Health and Safety; ISO 26000 - Social Responsibility, together with the ICH Good clinical practice E6(R2) and the WMA Declaration of Helsinki.
QUALITY STANDARDIZATION, SERVICE SUSTAINABILITY and PEOPLE WELLBEING is a challenging combination of our distinct value proposition on the market.
Image courtesy of fotographic1980 at FreeDigitalPhotos.net
All customers and consumers today ask for greater safety and more guarantees. For the sake of satisfying these specific requests and ensuring the correctness, sustainability and efficiency of our working processes, we observe in particular the following ISO Standards: ISO 14155 - Clinical Investigation of Medical Devices for Human Subjects; ISO 22000 - Food Safety Management; ISO 45001 - Occupational Health and Safety; ISO 26000 - Social Responsibility, together with the ICH Good clinical practice E6(R2) and the WMA Declaration of Helsinki.
QUALITY STANDARDIZATION, SERVICE SUSTAINABILITY and PEOPLE WELLBEING is a challenging combination of our distinct value proposition on the market.
Image courtesy of fotographic1980 at FreeDigitalPhotos.net
#SDG 2 Treatment of Anaemia | Food Supplement RUBENFLUX®
06/07/2020
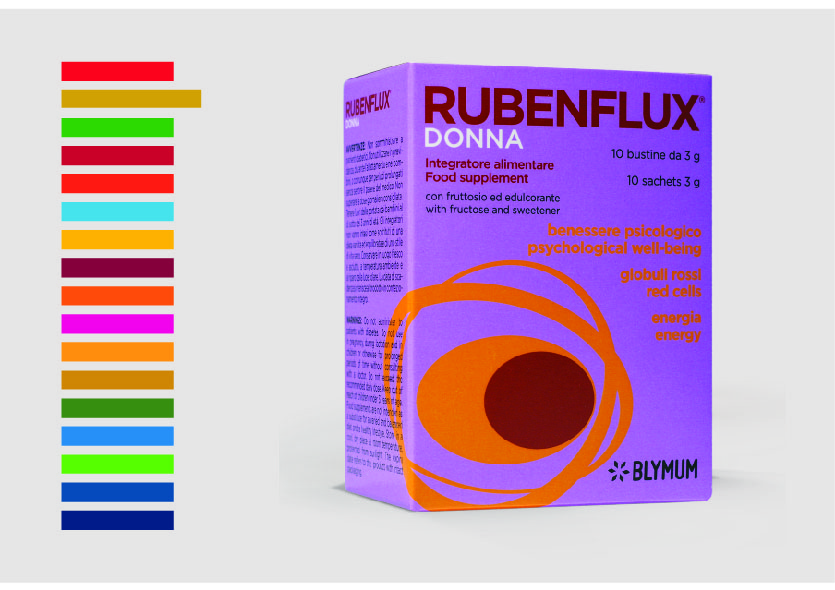
Inside the 2nd United Nations' SDG Goal: END HUNGER, ACHIEVE FOOD SECURITY AND IMPROVED
NUTRITION AND PROMOTE SUSTAINABLE AGRICULTURE, there is a particular Target 2.2, aimed at ending all forms of malnutrition, and addressing the nutritional needs of adolescent girls, pregnant and lactating women and older persons. Within the Global indicator framework for the Sustainable Development Goals and targets of the 2030 Agenda for Sustainable Development, a particular attention is given to a specific KPI “Prevalence of anaemia in women aged 15 to 49 years, by pregnancy status (percentage)”.
Our product RUBENFLUX® is a dietary supplement of essential amino acids, vitamin C, Group B vitamins and iron, for the female market, indicated for the red blood cells and hemoglobin natural formation and for the reduction of tiredness and fatigue. The RUBENFLUX® formulation, supported by clinical trials, scientific publications and granted with an IP by the Italian Patent office, is developed following the basis of Physiological Modulation.
RUBENFLUX® is proposed for on-demand production, international licensing and/or definitive purchase for those Companies strategically involved in the treatment of anaemia, directly or through commercial activities.
For further information/queries please contact: info@blymum.com
NUTRITION AND PROMOTE SUSTAINABLE AGRICULTURE, there is a particular Target 2.2, aimed at ending all forms of malnutrition, and addressing the nutritional needs of adolescent girls, pregnant and lactating women and older persons. Within the Global indicator framework for the Sustainable Development Goals and targets of the 2030 Agenda for Sustainable Development, a particular attention is given to a specific KPI “Prevalence of anaemia in women aged 15 to 49 years, by pregnancy status (percentage)”.
Our product RUBENFLUX® is a dietary supplement of essential amino acids, vitamin C, Group B vitamins and iron, for the female market, indicated for the red blood cells and hemoglobin natural formation and for the reduction of tiredness and fatigue. The RUBENFLUX® formulation, supported by clinical trials, scientific publications and granted with an IP by the Italian Patent office, is developed following the basis of Physiological Modulation.
RUBENFLUX® is proposed for on-demand production, international licensing and/or definitive purchase for those Companies strategically involved in the treatment of anaemia, directly or through commercial activities.
For further information/queries please contact: info@blymum.com
REGULATORY COMPLIANCE SERVICES | Smart Wellbeing Modulations
08/06/2020

Recently, we have drawn up and followed the submission, through an international recognized patent office, of a third party’s patent application pertaining to a Class III medical device to treat Obstructive Sleep Apnea (OSA). The patent application has successfully been published last July 2019 and is currently being finally approved and released by the European Patent Office (EPO) as an official intellectual property.
If new Clients wish us to assist them in studying, drafting and deposit new IPs, we will be proud to assist them with dossier filing, technical support, and regulatory compliance.
For more information contact: info@blymum.com
Image courtesy of Danilo Rizzuti at FreeDigitalPhotos.net
If new Clients wish us to assist them in studying, drafting and deposit new IPs, we will be proud to assist them with dossier filing, technical support, and regulatory compliance.
For more information contact: info@blymum.com
Image courtesy of Danilo Rizzuti at FreeDigitalPhotos.net
